|
Eligible patients were adults (>= 18 years) with previously untreated locally advanced or metastatic non-small-cell lung cancer without a sensitising EGFR mutation or ALK translocation and with an Eastern Cooperative Oncology Group (ECOG) performance status score of 0 or 1, life expectancy 3 months or longer, and a PD-L1 TPS of 1% or greater. Methods This randomised, open-label, phase 3 study was done in 213 medical centres in 32 countries. We investigated overall survival after treatment with pembrolizumab monotherapy in patients with a PD-L1 TPS of 1% or greater. LANCET, v.393, n.10183, p.1819-1830, 2019īackground First-line pembrolizumab monotherapy improves overall and progression-free survival in patients with untreated metastatic non-small-cell lung cancer with a programmed death ligand 1 (PD-L1) tumour proportion score (TPS) of 50% or greater. BONDARENKO, Igor KUBOTA, Kaoru LUBINIECKI, Gregory M. CASTRO JR., Gilberto SRIMUNINNIMIT, Vichien LAKTIONOV, Konstantin K. WU, Yi-Long KUDABA, Iveta KOWALSKI, Dariusz M. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial Treatment-related adverse events of grade 3 or worse occurred in 113 (18%) of 636 treated patients in the pembrolizumab group and in 252 (41%) of 615 in the chemotherapy group and led to death in 13 (2%) and 14 (2%) patients, respectively.Please use this identifier to cite or link to this item: As of Feb 26, 2018, median follow-up was 12♸ months. 599 (47%) had a TPS of 50% or greater and 818 patients (64%) had a TPS of 20% or greater. 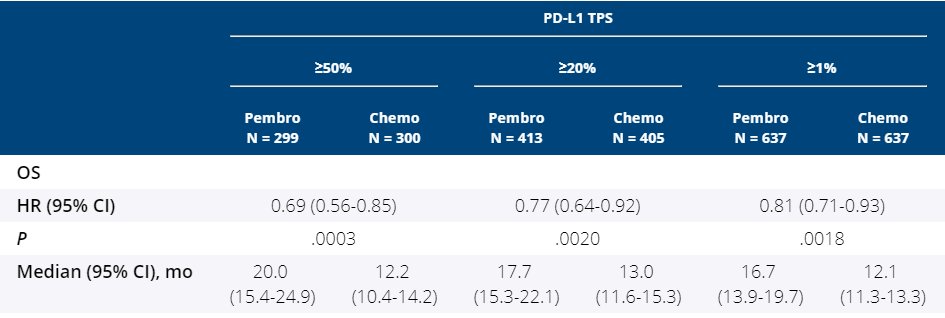
This study is registered at, number NCT02220894.įrom Dec 19, 2014, to March 6, 2017, 1274 patients (902 men, 372 women, median age 63 years ) with a PD-L1 TPS of 1% or greater were allocated to pembrolizumab (n=637) or chemotherapy (n=637) and included in the intention-to-treat population. Primary endpoints were overall survival in patients with a TPS of 50% or greater, 20% or greater, and 1% or greater (one-sided significance thresholds, p=0♰122, p=0♰120, and p=0♰124, respectively) in the intention-to-treat population, assessed sequentially if the previous findings were significant.   
Enrolled patients were randomly assigned 1:1 in blocks of four per stratum to receive pembrolizumab 200 mg every 3 weeks for up to 35 cycles or the investigator's choice of platinum-based chemotherapy for four to six cycles. Randomisation was computer generated, accessed via an interactive voice-response and integrated web-response system, and stratified by region of enrolment (east Asia vs rest of world), ECOG performance status score (0 vs 1), histology (squamous vs non-squamous), and PD-L1 TPS (≥50% vs 1-49%). Eligible patients were adults (≥18 years) with previously untreated locally advanced or metastatic non-small-cell lung cancer without a sensitising EGFR mutation or ALK translocation and with an Eastern Cooperative Oncology Group (ECOG) performance status score of 0 or 1, life expectancy 3 months or longer, and a PD-L1 TPS of 1% or greater. This randomised, open-label, phase 3 study was done in 213 medical centres in 32 countries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
